The Sadtler research group uses light to image and control chemical transformations in inorganic nanostructures.
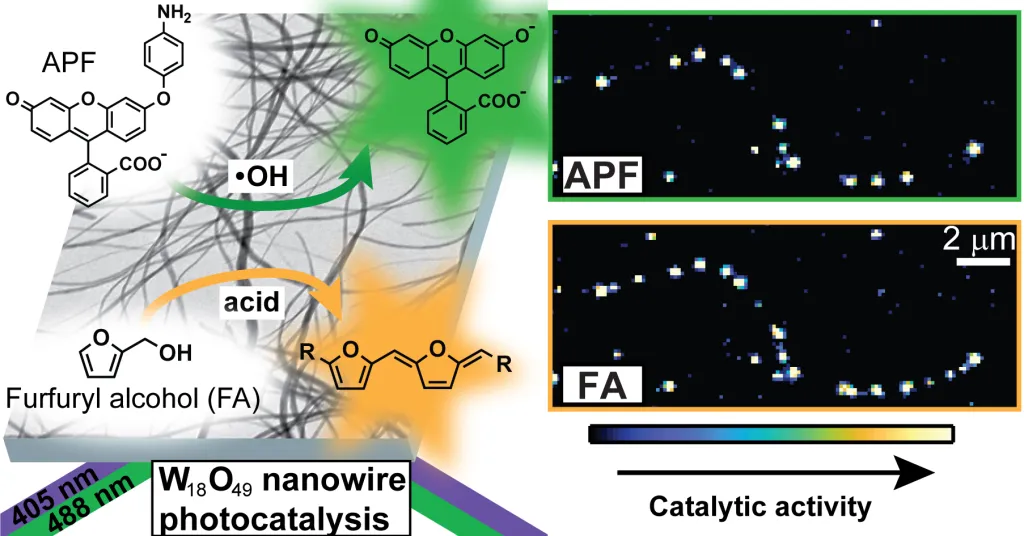
Imaging heterogeneity in the reactivity of semiconductor nanocrystals. Unlike molecular synthesis in which a single desired product can often be isolated, the synthesis of nanoscale particles inherently produces a distribution of sizes and shapes. We make measurements one particle at a time to understand how this heterogeneity impacts the chemical reactivity of colloidal semiconductor nanocrystals. Currently, we are using single-molecule fluorescence microscopy and chemically activated fluorogenic probes to map nanoscale spatial variations in the photocatalytic activity of semiconductor particles. These studies have provided new insights into how the concentration and distribution of oxygen vacancies control the activity of metal oxide semiconductors for fuel-forming reactions
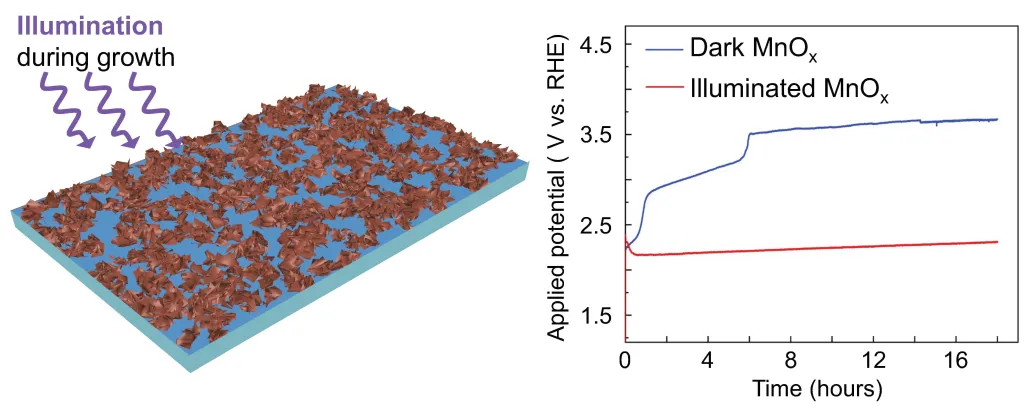
Growth of inorganic nanostructures under external stimuli. Chemists typically use parameters such as temperature, pressure, and concentration to direct chemical transformations in molecules and materials. Many classes of materials are also responsive to external stimuli, including light, electrochemical potential, and mechanical strain. We are designing adaptive inorganic materials that adjust their growth in response to multiple external stimuli to synthesize complex nanostructures and metastable materials that cannot be made by traditional synthetic methods.
Solution-phase synthesis of metastable compounds. The identity and arrangement of atoms in a crystal controls its electronic structure and resulting chemical and physical properties. Metastable phases have a structure with a local minimum in free energy, but which is not the thermodynamically most stable structure. Many metastable materials exhibit promising catalytic, electronic, and optical properties that make them attractive for applications in the conversion and storage of energy and information. However, it is still a fundamental challenge to develop general synthetic conditions that promote the formation of a metastable phase. We are developing solution-phase chemical routes to synthesize metastable phases of semiconductor compounds with unique optoelectronic properties for applications in nanophotonics and quantum computation.





